
Growing a nutraceutical or supplement brand brings new operational pressures long before it brings higher volume. Scaling a nutraceutical or supplement brand is primarily an operational challenge.
For procurement heads, operations managers, and founders, managing the supplement manufacturing process at scale is often the biggest barrier to growth. Inconsistent quality, CGMP gaps, or a single material failure can trigger recalls, increase scrap rate, and disrupt lead time stability within hours.
As the US dietary supplement market is projected to reach $124.22 billion by 2033, scaling requires precise control over all production phases beyond formula strength and branding.
This guide breaks down the seven core steps of the supplement manufacturing process and shows how each decision affects KPIs such as Scrap Rate, Yield Improvement, Equipment Uptime, Lead Time Stability, and Cost Per Unit.
Key Takeaways
Start with a stable formula that holds its potency and performs well during real-world manufacturing.
Use verified raw materials so your batch stays compliant, consistent, and free from supplier-related delays.
Blend ingredients evenly to avoid potency issues and keep downstream production running smoothly.
Form and package the product accurately to protect quality, reduce defects, and meet regulatory standards.
Test and approve each batch before shipment to maintain safety, traceability, and predictable lead times.
What is the Supplement Manufacturing Process?
The supplement manufacturing process, governed by the FDA's Current Good Manufacturing Practices (CGMP), transforms raw ingredients into a finished, shelf-stable, legally compliant product while ensuring batch-to-batch consistency and consumer safety.
In practice, this process is not a straight line. There are continuous feedback loops between R&D, Quality Control (QC), and Operations. Pilot batches may need reformulation. QC findings may send a product back to R&D. Operations might raise issues with flowability or run speed. The goal is to keep Throughput high without driving up Cost Per Unit or Scrap Rate.
With that big-picture view in place, the first operational checkpoint is a formula that can survive real-world manufacturing and shelf life.
Stats: The US dietary supplements market is expected to grow at a CAGR of 7.7% from 2025 to 2033, as per Grand View Research.
Also Read: How to Start a Skincare Line in 2025
Seven Steps in the Supplement Manufacturing Process
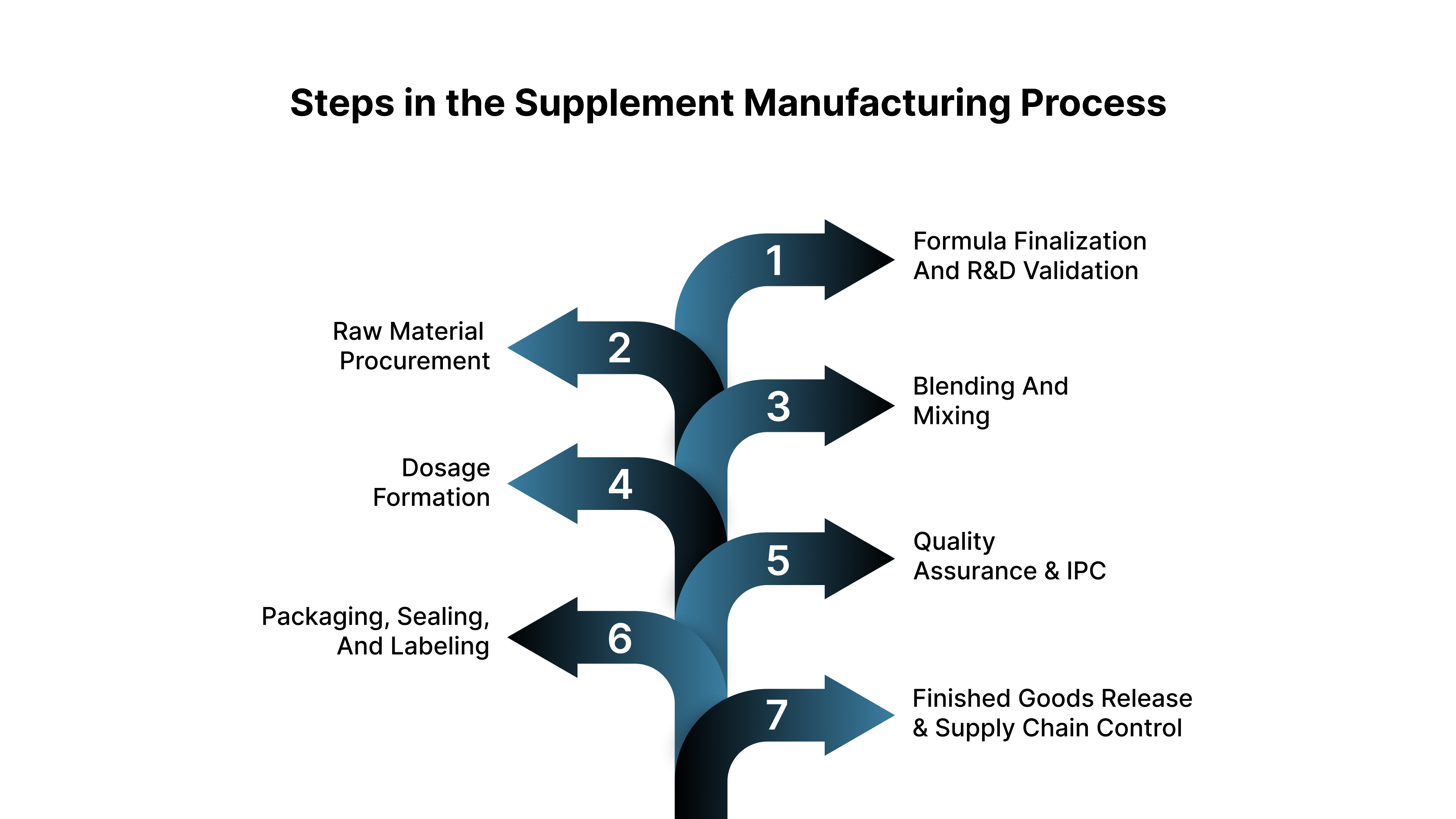
Below are the seven core stages every supplement batch must pass to remain compliant, consistent, and releasable.
Step 1: Formula Finalization and R&D Validation
This pre-production phase locks the master formula. Here, the team decides:
Final ingredient ratios
Choice of excipients (binders, fillers, flow agents)
Required overages to keep label potency at expiry
Preferred dosage form and target organoleptic properties
Pilot runs and stability studies test how the formula behaves in real conditions, not just in a spreadsheet.
Why It Matters
Yield Improvement: A stable formula minimizes potential waste during the initial pilot runs.
Stability Testing: Successful validation here ensures a 2-3 year shelf life, protecting the brand's inventory investment and minimizing Scrap Rate from spoilage.
What to Look Out For (Challenges)
Ingredient Incompatibility: Active ingredients reacting with excipients or capsule shells (e.g., moisture sensitivity causing hardening). This issue will halt the supplement production process immediately during pilot testing.
Flowability Issues: Powders that clump or do not flow smoothly can fail to dose correctly in high-speed machines, compromising Content Uniformity and destroying the batch.
Once the master formula is locked and validated, attention shifts to sourcing inputs that can meet those specifications at scale.
Step 2: Raw Material Procurement and IQC Vetting
Procurement and Quality source and vet active ingredients, excipients, and all packaging components. Vendors provide Certificates of Analysis (CoAs), which are then verified by incoming quality control (IQC) through identity, purity, and safety testing.
Why It Matters
Lower Supplier Risk: Qualified vendors reduce variability and prevent production stoppages.
Compliance Support: Materials that meet microbial, heavy metal, and purity limits protect CGMP compliance.
Lead Time Stability: Reliable suppliers keep inputs available and production schedules predictable.
What to Look Out For (Challenges)
Identity Failure: Incoming materials that do not match the CoA must be rejected, causing delays and downtime.
Packaging Mismatch: Bottles or caps that fail dimensional Acceptable Quality Level (AQL) checks cannot run on automated lines.
Once materials are approved and released from quarantine, they move to blending, where content uniformity becomes the next critical risk.
Step 3: Blending and Mixing
Blending combines all ingredients into a uniform mixture using CGMP-compliant stainless steel equipment such as V-blenders, ribbon blenders, or double-cone blenders. Loading pattern, blend time, and speed follow validated procedures.
Why It Matters
Content Uniformity: A consistent blend ensures accurate dosing; failures often require batch destruction.
Throughput: Efficient blending keeps downstream equipment running at planned speeds.
What to Look Out For (Challenges)
Segregation: Particle size or density differences cause potency variation and raise the Scrap Rate.
Cross-Contamination: Residues from previous batches can contaminate the blend if cleaning isn't thorough.
Once blending passes checks, the batch moves to encapsulation, tableting, or filling.
Step 4: Dosage Formation (Encapsulation, Tableting, or Filling)
In this step, the blended powder is converted to its final format:
Two-piece hard capsules
Compressed tablets
High-speed machines dose, compress, or fill product according to validated set-ups and standard operating procedures, which ensure precise fill weights and regulatory compliance.
Why It Matters
Fill Accuracy: Tight weight control protects compliance and prevents inflated Cost Per Unit.
Equipment Uptime: Validated, well-maintained machines keep runs stable and support lead time stability.
What to Look Out For (Challenges)
Capsule/Tablet Splitting: Sticky blends or incorrect pressure cause defects, line stoppages, and a higher Scrap Rate.
Low Disintegration: Tablets that don't break down quickly risk failing performance tests and compliance checks.
Once dosage units are formed, QA performs verification to confirm they match the label and master formula before proceeding to packaging, making sure of product integrity.
Worried about which packaging format will fit best with your production line? Connect with Respect Manufacturer for an all-in-one solution for your dietary supplements, skincare, and personal care products.
Step 5: Quality Assurance and In-Process Checks (IPC)
Quality teams run in-process checks during and after Step 4, followed by finished product testing, often through external labs.
Typical checks include:
Capsule or tablet weight and weight variation
Disintegration time
Torque and seal checks (for packaging trials)
Appearance and defect inspection
Finished product testing usually covers:
Potency versus label claim
Impurities and related substances
Microbial limits and pathogens
Heavy metals, if relevant
Why It Matters
Batch Record Integrity: All IPC and lab data must be documented to confirm CGMP compliance.
Risk Prevention: Early detection of issues prevents costly batch failure later.
What to Look Out For (Challenges)
Out-of-Spec Potency: May require full batch rework or destruction.
Microbial Contamination: Stops release and triggers facility-level investigation.
Step 6: Packaging, Sealing, and Labeling
The formed dosage units are counted, filled into the primary packaging (bottles, jars, stickpacks), sealed (induction seal, cap), labeled, and unitized into secondary packaging (boxes).
Why It Matters
Protection and Longevity: Correct induction sealing and capping protect the product from moisture and air, preserving the Stability Testing results and extending shelf life.
Compliance: Labels must be correctly applied, legible, and match the final approved proof, including correct lot numbers and expiration dates. Label errors are a leading cause of FDA warning letters.
What to Look Out For (Challenges)
Cap Torque Failure: The cap is sealed too loosely or too tightly. This fails to protect the product or frustrates the consumer, increasing customer returns.
Label Misalignment: Labels are applied incorrectly or misregistered, which requires immediate line stoppage and manual correction, increasing Downtime Reduction.
After packaging, the product appears ready to ship, but it cannot move to customers until released by the Quality Unit.
Step 7: Finished Goods Release and Supply Chain Control
This stage moves the packaged batch from Quarantine to Released status. The Quality or Regulatory Manager signs the Batch Record after verifying that all tests, deviations, and documentation meet the master formula and CGMP requirements.
Why It Matters
Product Traceability: Every unit links back to its raw material CoAs and production data, enabling fast, targeted recalls.
Lead Time Stability: A consistent release process keeps shipment dates predictable and prevents penalties.
What to Look Out For (Challenges)
Documentation Gaps: Missing signatures, incomplete deviations, or unverified calibrations block release and add operational complexity.
Labeling or Lot Errors: Mismatched lot numbers or incorrect expiry dates trigger rejection and costly rework.
CGMP underpins this entire process. Understanding FDA expectations helps keep each batch releasable and compliant.
How Does the FDA Take Part in the Supplement Manufacturing Process?
The FDA does not approve dietary supplements before they reach the market in the way it approves drugs. Instead, it enforces CGMP regulations to control how supplements are manufactured, packed, and held.
FDA Role | What It Covers | Why It Matters Operationally |
CGMP (21 CFR Part 111) | Standards for raw materials, manufacturing controls, documentation, and QA/QC. | Prevents batch failures and supports consistent, compliant production |
Inspections & Audits | Reviews batch records, deviations, sanitation, and quality systems. May issue Form 483s or warning letters. | Requires complete documentation and disciplined processes to avoid production delays |
Adverse Event Reporting | Mandatory reporting of serious consumer adverse events. | Poor reporting can trigger compliance actions and brand risk. |
Labeling & Claims Control | Allows structure/function claims; prohibits disease claims. Requires accurate Supplement Facts. | Incorrect labels lead to rework, holds, or recall risk. |
Note: FDA oversight shapes how supplements are made, documented, and released. Weak processes increase compliance costs and disrupt profitability.
These expectations add structure to the supplement manufacturing process. They also add cost when operations are inefficient, which is why profitability depends so heavily on day-to-day execution.
Also Read: The Ultimate MOCRA Guide 2025: FDA Requirements & Timelines
Is Supplement Manufacturing Profitable?
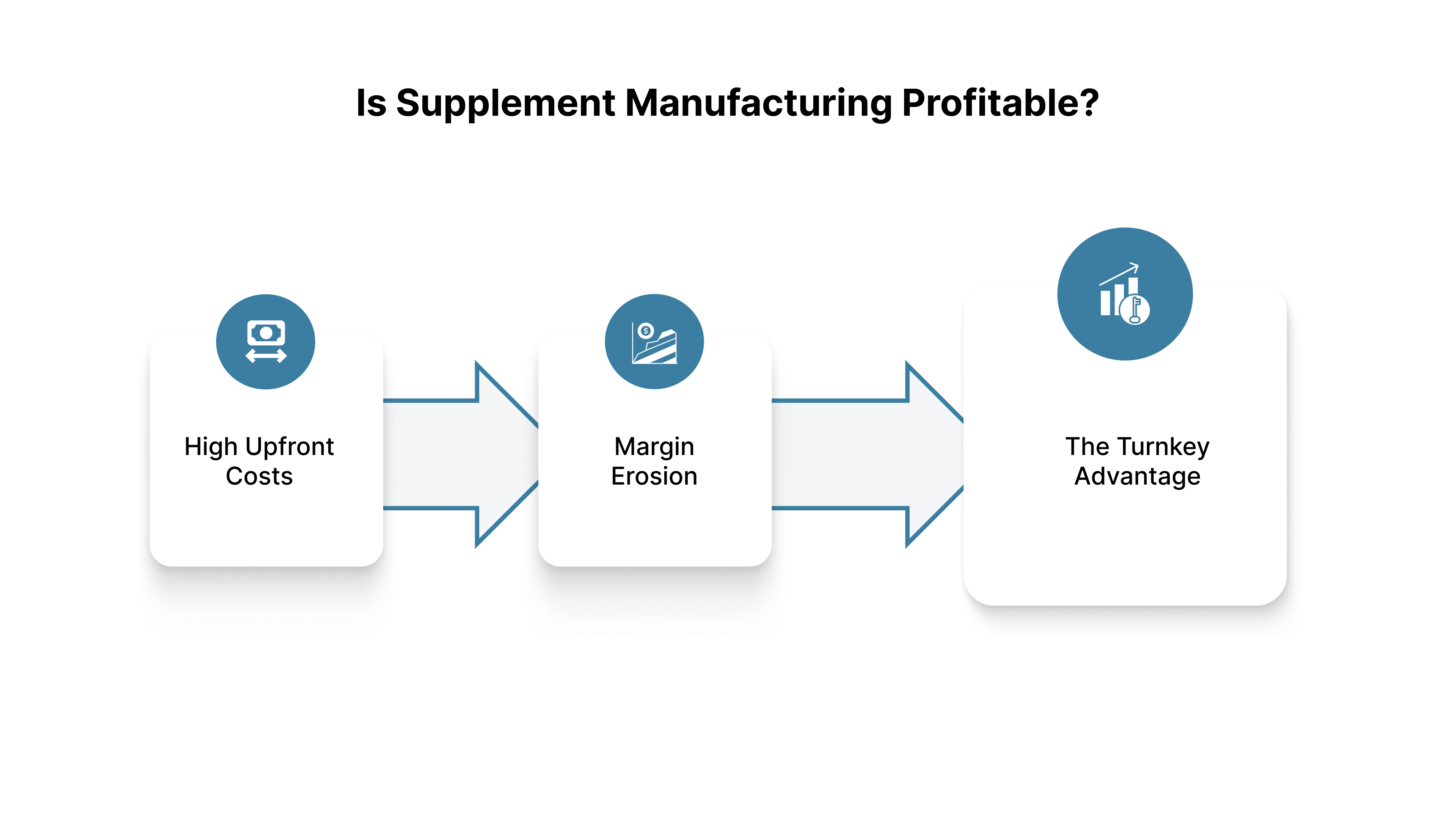
The supplement industry is highly profitable, but margins are eroded by operational inefficiencies. Profitability depends entirely on controlling the Cost Per Unit (CPU) and maintaining production Throughput.
High Upfront Costs: Initial profitability is constrained by high costs for stability testing, CGMP compliance, R&D, and Minimum Order Quantities (MOQs).
Margin Erosion: The greatest threat to long-term profitability is batch failure. Any batch that fails Potency Testing or requires extensive rework due to poor blending immediately spikes the CPU and destroys profit margins for the entire production run.
The Turnkey Advantage: By consolidating the process with a turnkey manufacturer, brands reduce indirect costs associated with managing multiple vendors, shipping component materials, and dealing with quality failures across different sites, which helps stabilize long-term profitability.
To realize those benefits, you still need a structured way to select the right partner for your supplement manufacturing process.
How to Choose the Right Supplement Process and Partner?
Choosing the right supplement production process is less about the lowest bid and more about guaranteed quality and reliable scale. The strategic choice lies in selecting a partner that de-risks the entire journey.
Selection Criteria | Operational Question | Risk Mitigation |
Compliance Proof | Is the manufacturer currently FDA-registered and provably CGMP-certified? | Avoids regulatory shutdowns and product seizures. |
Material Control | Does the facility perform in-house Identity Testing and strict AQL on incoming materials? | Eliminates Supplier Risk of receiving counterfeit or contaminated ingredients. |
Scale Capability | Do they use automated stainless steel lines capable of high-volume Throughput for your dosage form? | Helps production scale without sacrificing quality or increasing Cost Per Unit. |
Logistics Model | Do they offer a turnkey solution that handles R&D, compliance, and logistics? | Reduces Operational Complexity and guarantees better Lead Time Stability. |
Once you define what "good" looks like across these areas, you can compare manufacturers beyond price. Many brands then prefer a turnkey partner that integrates these capabilities in one place.
Partnering with Respect Manufacturing: A Turnkey Solution
For fast-scaling supplement brands, the most effective strategy to minimize risk and accelerate growth is to utilize a single, dedicated manufacturing partner. Respect Manufacturing's turnkey model directly addresses the complexity and operational risks outlined in these seven steps.
Respect Manufacturing's Operational Advantages
Here is how a turnkey partner like Respect Manufacturing can address common operational risks and KPI concerns:
Operational Risk / KPI Focus | Respect Manufacturing Solution | Practical Impact for Your Team |
Quality inconsistency across batches | Centralized QC, IQC testing on all inputs, and stability programs for key SKUs. | Lower Scrap Rate and fewer reworks from potency or contamination failures. |
Supplier risk and material variability | Qualified raw material and packaging suppliers, plus AQL checks on incoming components. | Reduced supplier risk and more predictable lead time stability. |
Lead time volatility | Integrated scheduling of formulation, production, packaging, and testing. | More reliable on-time shipments and fewer emergency schedule changes. |
High CPU due to multi-vendor setup | Single facility for R&D, manufacturing, and packaging, with aligned batch sizes and formats. | Lower indirect costs from freight, handoffs, and repeated audits. |
Operational complexity for new launches | Support across formula refinement, packaging selection, and stability planning. | Faster launch timelines and smoother SKU expansions. |
By working with Respect Manufacturing, which internalizes these quality, testing, and scheduling responsibilities, your internal team can focus on product strategy, channel expansion, and brand building, while still holding clear KPIs across yield improvement, throughput, and lead time stability.
Stop managing multiple vendors and compliance risks. Optimize your supplement production today. Contact Respect Manufacturing to discuss your next supplement batch.
Also Read: 8 Simple Steps To Start A Nutrition Supplement Business
Conclusion
The supplement manufacturing process is a chain of operational decisions that directly affects scrap rate, yield improvement, equipment uptime, lead time stability, and cost per unit. Managing each step, from formula validation to material sourcing, blending, dosage formation, testing, packaging, and release, helps protect compliance and profitability as you scale.
For fast-growing wellness and nutraceutical brands, working with a turnkey partner can optimize these steps and reduce operational risk without adding complexity. Respect Manufacturing supports this approach by integrating formulation, production, testing, and packaging under one CGMP framework, helping brands achieve more predictable operations and reliable batch performance.
Contact us to take the next step toward a smoother, more predictable manufacturing process.
FAQs
1. What determines whether a supplement formula is ready for manufacturing?
A formula is ready when stability, compatibility, and overage requirements are validated through pilot runs and early QC testing.
2. How does raw material quality impact production efficiency?
Clean, verified inputs with accurate CoAs reduce downtime, prevent rejection at IQC, and support consistent batch performance.
3. Why is blending considered a high-risk step in the process?
Even small variations in particle size or density can cause segregation, leading to potency drift and increased Scrap Rate.
4. What role do Batch Records play during the release stage?
They provide documented proof that all tests, deviations, and equipment checks were completed according to CGMP before goods ship.
5. How can brands maintain predictable lead times during scale-up?
By using a partner with integrated formulation, production, testing, and packaging capabilities, reducing handoff delays between vendors.


